Chemical Information
| CAS Number | 68497-62-1 |
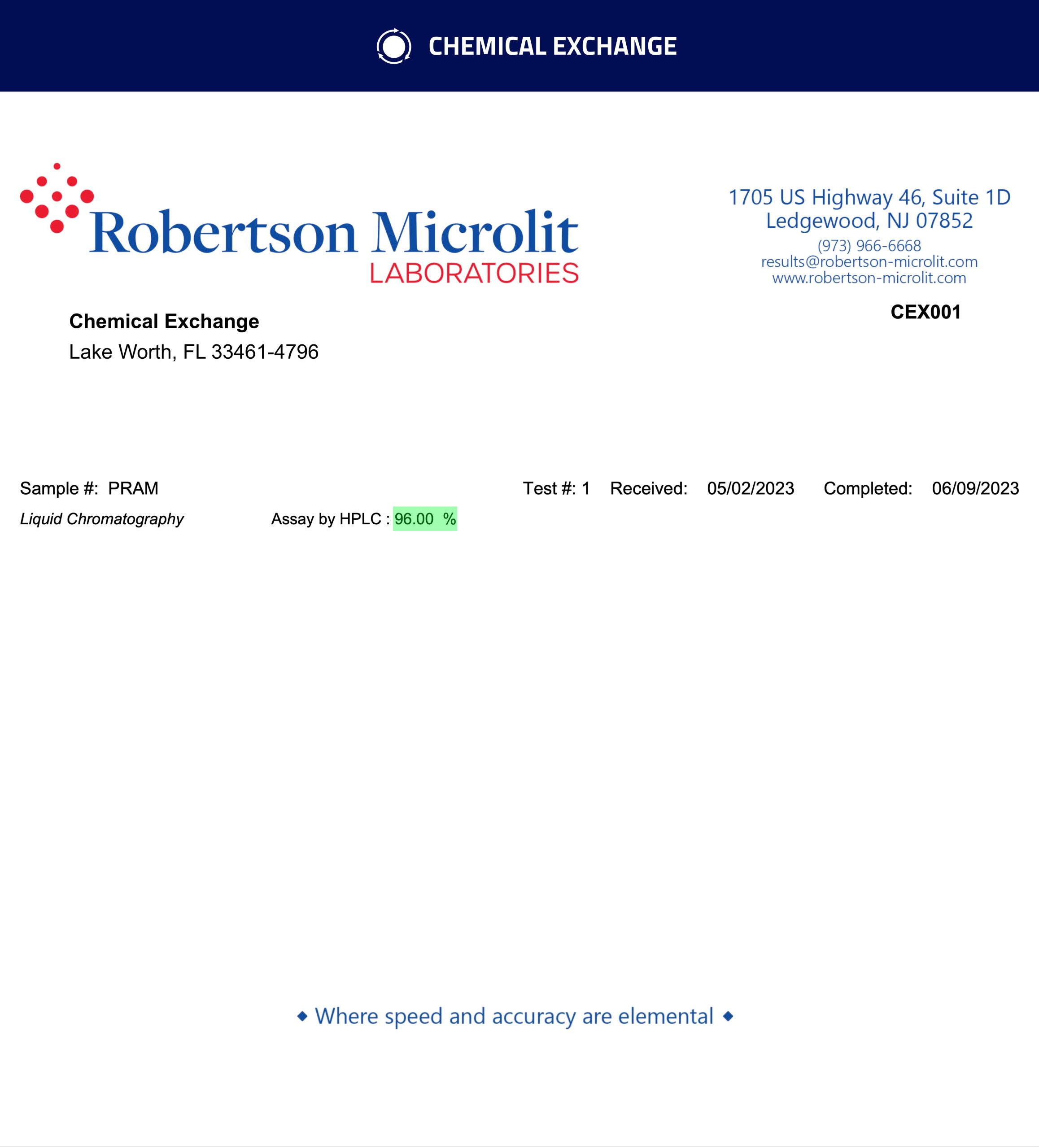
| Purity | 96.0% |
| Molecular Weight | 269.38 g/mol |
| Molecular Formula | C12H14N2O2 |
| Synonyms | 68497-62-1, 1-Pyrrolidineacetamide, N-(2-(bis(1-methylethyl)amino)ethyl)-2-oxo-, Remen, Neupramir, Pramistar |
| PubChem CID | 51712 |
| Smiles | O=C1N(CC(=O)NCCN(C(C)C)C(C)C)CCC1 |
Technical Information
| Application | Pramiracetam is a nootropic agent belonging to the racetam family of drugs, which is several-fold more potent than piracetam in research studies. |
| Appearance | White or off-white powder |
| Physical State | Solid |
| Solubility | Soluble in water (10 mg/ml), DMSO (54 mg/ml at 25 °C), and ethanol |
| Storage | Store at room temperature or cooler, in a sealed airtight container, protected from heat, light and humidity. |
| Stability | Stable for at least two years when stored as above. |
Biochemical Activity
Pramiracetam (Diisopropyl-(2-oxopyrrolidin-1-yl)acetamide) is a Nootropic agent belonging to the racetam family of drugs. It is structurally similar to Piracetam and was first synthesized in 1984 as a more potent derivative of the parent compound, Pircetam. Pramiracetam is used in Eastern European countries and has the brand names Remen (Parke-Davis), Neupramir (Lusofarmaco), or Pramistar (Firma).
It is not approved for any use by the US Food and Drug Administration or by the European Medicines Agency. Pramiracetam has been studied as a potential treatment for memory loss associated with Alzheimer’s disease and for cognitive problems resulting from brain injury.
Pramiracetam is absorbed well in humans and animals and has a prolonged, yet variable half-life. In humans, peak plasma concentrations (Cmax) are close to 2.71µg/mL for 400mg oral treatment, around 5.4µg/mL for 800mg, and closer to 8.98µg/mL for a 1600mg administration.
Studies using animals have noted a half-life of around 2.3-3.9 hours and effective absorption in the brain[2], while human studies indicate a longer half-life of between 2 to 8 hours.
Hippocampal cell studies indicate that Pramiracetam is ineffective in inducing changes in hippocampal cells in vitro. Similarly, studies have indicated a failure to significantly influence adrenergic receptors in the serotonergic, dopaminergic, GABAergic, muscarinic, and adenosinic receptors.
Interestingly, studies have shown that adrenalectomy (removal of one or both of the adrenal glands) inhibits the effects of Pramiracetam. This indicates the role of the adrenal glands in Pramiracetam’s mechanism of action.
Furthermore, studies have pointed out that drugs like aminoglutethimide and epoxymexrenone are able to inhibit Pramiracetam’s effects. These drugs are involved in the conversion of cholesterol to pregnenolone and aldosterone inhibition, respectively. These results indicate the role of steroidal hormones in Pramiracetam’s mechanism of action. Additional studies have indicated that raised corticosteroid levels have a limiting effect on the efficacy of racetam drugs, in general.
References
[1] Dutta A. (2015). “Ketamine and other potential glutamate antidepressants”. Psychiatry Res. 225(1-2):1-13. doi: 10.1016/j.psychres.2014.10.028. PMID: 25467702.