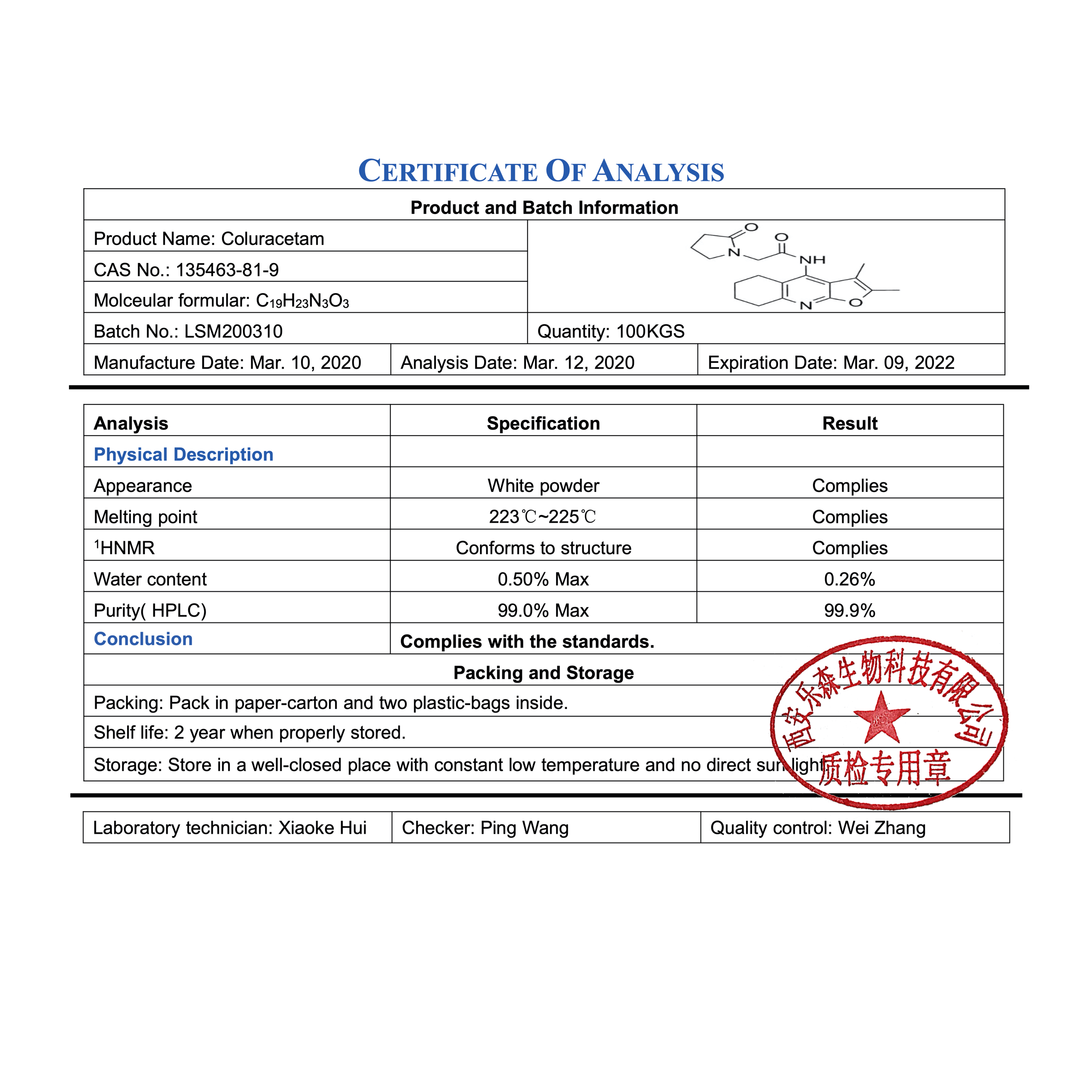
Chemical Information
| CAS Number | 135463-81-9 |
| Purity | 99.6% |
| Molecular Weight | 341.40 g/mol |
| Molecular Formula | C19H23N3O3 |
| Synonyms | N-(2,3-dimethyl-5,6,7,8-tetrahydrofuro[2,3-b]quinolin-4-yl)-2-(2-oxopyrrolidin-1-yl)acetamide, MKC-231 |
| PubChem CID | 214346 |
| Smiles | O=C1N(CCC1)CC(=O)NC2C4C(NC3C2CCCC3)OC(C4C)C |
Technical Information
| Application | Coluracetam (BCI-540) is a nootropic compound of the racetam family. |
| Appearance | White or Off-White Powder |
| Physical State | Solid |
| Solubility | Soluble to 50 mM in Ethanol, insoluble in Water. |
| Storage | Store at room temperature or cooler, in a sealed airtight container, protected from heat, light and humidity. |
| Stability | Stable for at least two years when stored as above. |
Biochemical Activity
Coluracetam (BCI-540, MKC-231) is a synthetic Nootropic drug in the racetam family that was initially developed by Mitsubishi Tanabe Pharma Corporation and later by Brain Cells Inc.
It was initially developed as a potential clinical therapy for Alzheimer’s Disease, but after trials failed to reach endpoints it was developed further for the treatment of major depressive disorder (MDD). Brain Cells Inc. reached phase 2a clinical trials which suggested that it may have potential as a medication for comorbid MDD and generalized anxiety disorder (GAD).
Animal studies have shown post administration (10mg/kg) plasma concentrations of 4.289+/-0.641µg/mL (30 minutes), 0.717+/-0.350µg/mL (3 hours), and 0.037+/-0.010µg/mL (24 hours). After 14 days of use, there did not appear to be any effects on pharmacokinetics, indicating no tolerance build-up.
Like other racetam compounds, Coluracetam has AMPA potentiating activity. Unlike most other racetams, it was additionally designed to activate the cholinergic system. It is therefore of interest to researchers involved in studies regarding the choline transport system, protection against cognitive impairment, and neurochemical pathways for development of novel antidepressants.
Coluracetam has cholinergic activity, significantly enhancing high-affinity choline uptake (HACU) – known to be the rate-limiting step of Acetylcholine (ACh) synthesis. BCl-540 increases Vmax (1.6fold) of HACU as well as Bmax (1.7fold) of [3H]-HC-3 binding, with strong affinity for high-affinity choline transporters (CHT1).
The enhancement of HACU velocity is of great importance to researchers in the fields of Alzheimer’s and other dementias. A variety of research has pointed to perturbation in HACU activity in Alzheimer’s patients. Preliminary evidence shows that the HACU activity-enhancing effects of BCl-540 may limited to subjects with malfunctioning acetylcholine systems, without significant effects on otherwise healthy subjects.
In 1998, Akaike et al. published results indicating that Coluracetam has the potential to help protect the brain from glutamate neurotoxicity induced by ionomycin, through the suppression of NO synthesis triggered by increased Ca2+ influx. However, the study used a concentration higher than normal for the molecule (10µM), indicating that the results may not be practically relevant.
References
[1] Dutta A. (2015). “Ketamine and other potential glutamate antidepressants”. Psychiatry Res. 225(1-2):1-13. doi: 10.1016/j.psychres.2014.10.028. PMID: 25467702.